Electrolysis (& Corrosion...)
Electrolysis
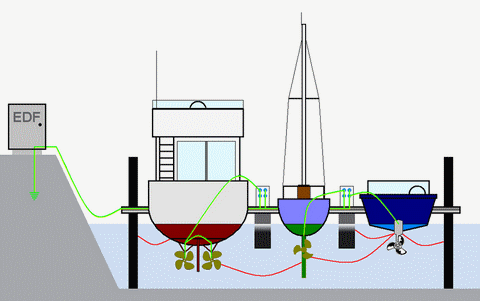
The term "electrolysis", which is a misnomer of the maritime community, refers here to natural galvanic and accidental electrolytic corrosion. Corrosion of metals is a complex and multiform electro-chemical phenomenon: it implies electrical natural and/or unwanted detectable currents and is a matter of concern for crews safety as well as money value of concerned boats.
Corrosion
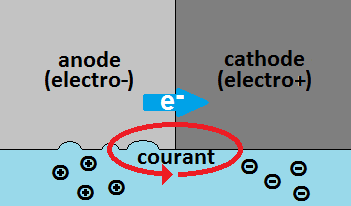
Corrosion is the result of an electrons movement (thus an electric current...), natural and/or accidental, between an anodic metal surface (i.e. your zinc anodes) and a cathodic metal surface (i.e. your aluminium sail-drive's leg), conductively linked and immersed in the same electrolyte (i.e. sea water). There are loss of material at the anode and protection at the cathode, hence the term "cathodic protection".
Galvanic vs electrolytic
Galvanic corrosion is due to natural potential differences between different metals in contact in a same electrolyte (conductive water...), which generates a very low intensity electrical current: the corrosion current. It is effectively adressed by sacrificial anodes which deliver a protection current, opposing the corrosion current. Attention, cathodic protection such obtained must not be presumed but measured!
Electrolytic corrosion, on the other hand, is induced by an additional and accidental direct current: the stray current, which amplify in a way or another - according to polarity - the effects of the corrosion current. Attention, harmful stray currents from on board or ashore always seek going to "earth" and are responsible for accelerated corrosion of the conductive metal at the outgoing point to sea! Anodes are of no use in this case and only early detection and quick elimination will preserve your boat from costly consequences!
The corrosion potential: an essential indicator!
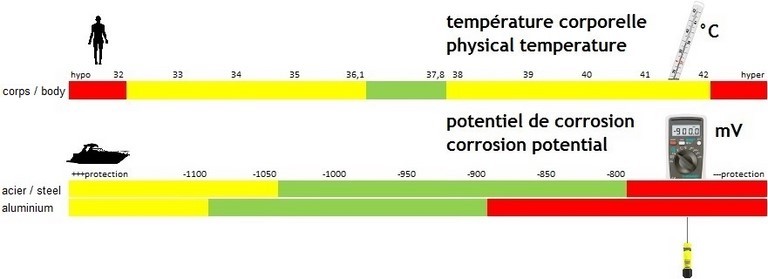
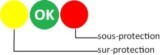
Corrosion protection of immersed metals is conjunctivally performed by their natural oxidation layer and/or by the paint system supplemented with available cathodic protection (anodes...). Efficiency depends upon achieved value of their corrosion potential, expressed in (-)mV. Like human physical temperature, control is done with a dedicated device, through a simple measurement with instantaneous understanding: corrosion potential must remain within protective range (green zone above). Value shows protection level given by anodes, which are in their key role, and warns you about eventual harmful stray currents from on board or ashore.
Over and under-protection: warning!
In a same metal an anode area always destroys itself in favor of a cathode area which is thus protected. When such areas are deliberatly created by linking together different metals (sacrificial anodes) or other means, this is then called "cathodic protection".
Sacrificial anodes must always deliver the right amount of protection current to counteract the corrosion current, and be replaced when inadequate. According to polarity, stray currents may cause over- or under- cathodic protection situations.
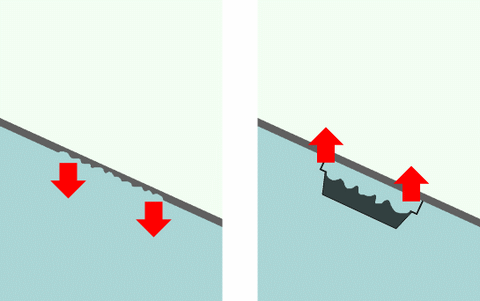
Under cathodic protection implies more or less corrosive attacks on steel (above strip n°2) and aluminium (above strip n°3). It happens when corrosion potential is higher than the maximum requested value. Over cathodic protection may cause bubling and disbonding of underwater paints (above strip n°1), if not more serious damages. It happens when corrosion potential is lower than the minimum requested value (algebraic meaning).
These may, of course, affects safety of navigation and result in costly maintenance operations or parts replacement.
Involvment of alternative AC current:mind the earthing wire!
Involvment of alternative current has not been scientifically proven concerning corrosion of immersed metal equipments of ships. However, the earthing wire of 230 V shore power might carry direct stray currents, potentially very dangerous for your immersed metal equipments if, as requested by safety regulations, it is connected to the boat's earth. This promotes indeed a corrosive loop, necessary for corrosion to occur. As you can never guess quality of shore or neighbourhood installations, prevention goes through regular control of corrosion potential and availability of appropriate protection devices: galvanic isolators or isolation transformers.![]() It's no use tripping breakers to keep safe: the earth wire by-pass all switches...
It's no use tripping breakers to keep safe: the earth wire by-pass all switches...
The White Paper about cathodic protection...

Regularly supplemented and updated, the actual edition includes some attacks commonly encountered on oxido-protected metals: stainless steel, aluminium and bronze.
Anodes
Anodes deliver a protection current opposing the corrosion current. If it is not sufficient, then problems occurs... Zinc, Aluminium or Magnesium, it is more the buyer's problem to know what he buys than the seller's one to know what he sells!
Isolator or transformer?
Galvanic isolators or isolation transformers stop stray currents from "getting" onboard through the shore earth wire. They imply a different technology, reliability and effectiveness and their relative price vary from 1 to 10.
Paint blistering
In addition to over-protection, a common cause for immersed paints blistering of a steel hull, and particularly in fresh water, is the osmotic pressure difference due to indesirable soluble impurities beneath the paint layer.